|
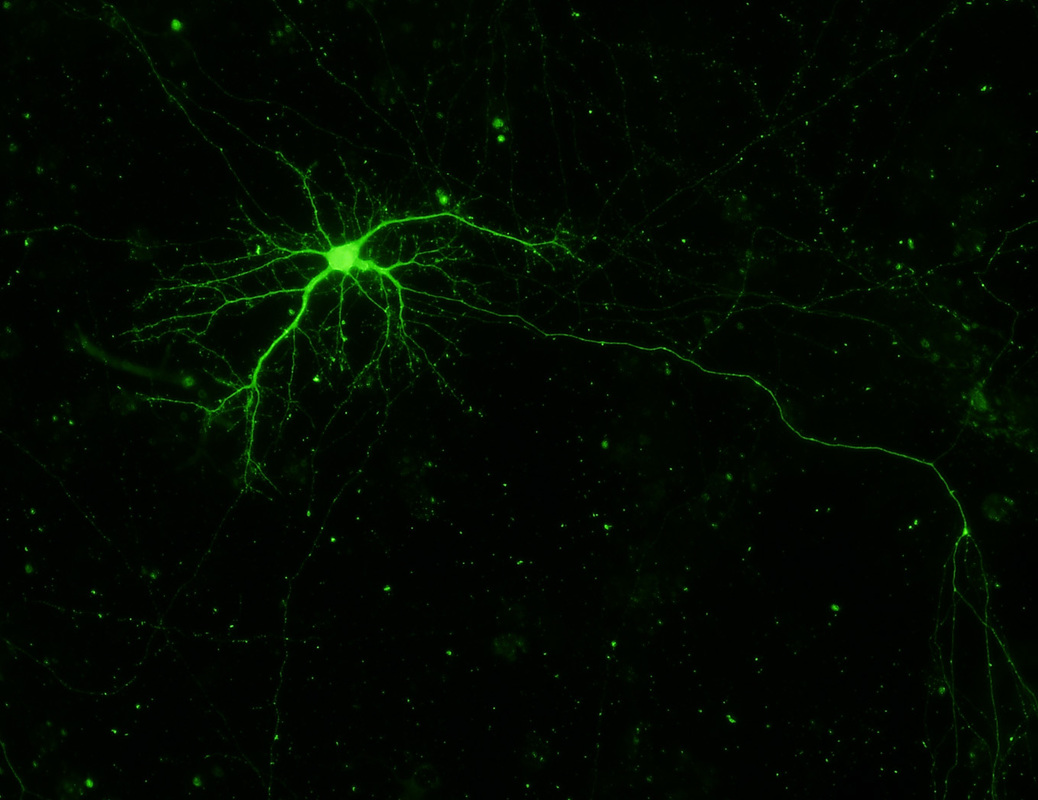
Regulation of dendritic arborization and synaptic structures. Autosomal recessive mutations in PTEN-induced putative kinase 1 (PINK1) cause a familial form of Parkinson's disease, with executive dysfunction and neuropsychiatric changes in heterozygote carriers. Autosomal dominant mutations in leucine-rich repeat kinase 2 (LRRK2) represent the most common known cause of PD. Mutations in both lead to attenuated dendritic arbors and mitochondrial calcium dysregulation. Using unbiased methods, we identified valosin-containing protein (VCP) as a key partner in PINK1-mediated dendritogenesis. Current efforts are centered on understanding how mutations in these proteins result in dendritic degeneration and loss of mature spines.
|
|
Mitochondrial biogenesis, transport and mitophagy. We discovered that the ability to regenerate mitochondria plays a key role in determining whether mitophagy is adaptive or maladaptive. A key area of interest concerns dual function macromolecules, traditionally associated with mitochondria, but with additional signaling functions in the nucleus or cytosol. We have created new iPSC-based models to study brain and liver dysfunction in POLG-mutated syndromes. As POLG encodes the mitochondrial DNA polymerase, which is expressed in every cell, we are interested in cell-type specific compensation for impaired POLG function and what triggers acute decompensation in children with these diseases. Interestingly, mutations in POLG have also been linked to early-onset familial parkinsonism.
|
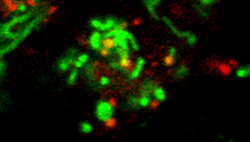
Proteostasis in Neurons. Maintaining protein homeostasis is challenging for neurons as a high demand for protein synthesis, folding and transport generates a constant source of stress. Evidence of protein mishandling is observed in nearly all neurodegenerative diseases. Autophagy, or "self-eating" plays an important role in the surveillance of long lived proteins and organelles, removing damaged structures for recycling. On the other hand, dysregulated autophagy contributes to dendritic shrinkage and loss of dendritic mitochondria. The AAA-ATPase valosin-containing protein (VCP) plays a central role in maintaining multiple aspects of protein homeostasis, including proteasomal and autophagic degradation and nuclear, ER and Golgi function. Mutations in VCP have been linked to several forms of frontotemporal dementia with or without concurrent musculoskeletal dysfunction, whereas mutations in tau are linked to other forms of frontotemporal dementia. We have created iPSC models from patients with familial neurodegeneration and are studying the potential role of disrupted proteostasis in neurodegeneration.
If you are interested in autophagy in neurons in general, or the role of autophagy in other brain disorders, please check the Neuronal Autophagy site.