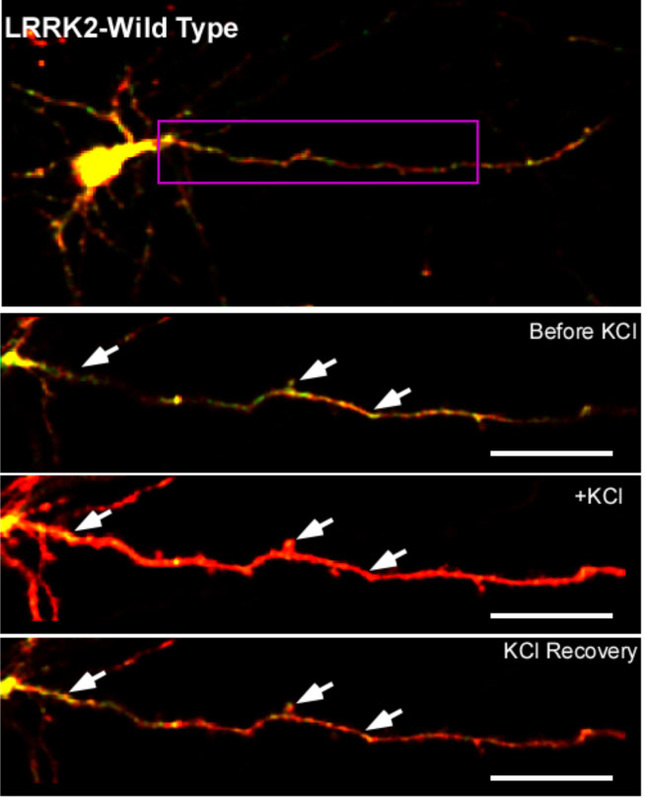
Lab alum Sam Cherra (front left) reports the funding of his R01 entitled Regulation of synapse development by small GTPase cascades in Caenorhabditis elegans. He was previously funded on an K99/R00 to study the excitation/inhibition balance in neural circuits. As a former graduate student, he not only continues his successful history of funding stemming back to his F31 on PINK1 and LRRK2 in the Chu Lab, but also has successfully mentored his own graduate student through the grant-writing process! Congratulations again.
0 Comments
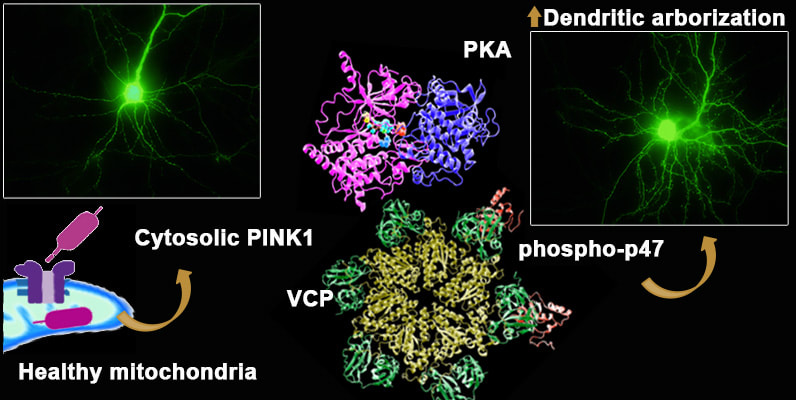
Our study showing that the recessive PD gene PINK1 plays an important role in regulating dendritic branching and synaptic structure and function is out as an early release! https://www.jneurosci.org/content/early/2022/09/02/JNEUROSCI.0785-22.2022 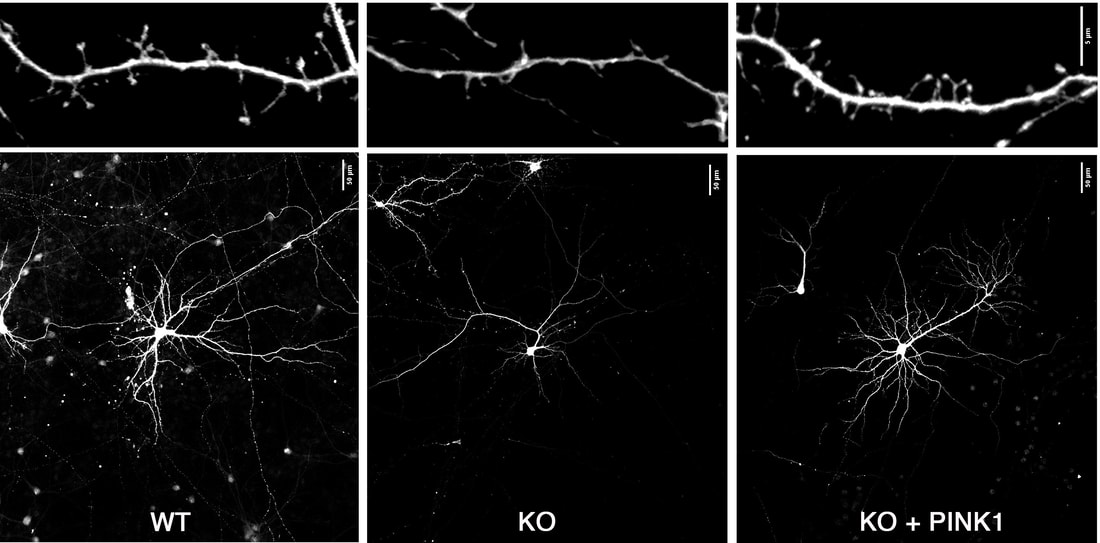 Click here to read this blog post by a science writer, which offers a great summary directed at students and non-scientist readers!
We show for the first time that endogenous PINK1 is essential for proper development of key neuronal structures responsible for receiving messages from other cells. The dendrites are like telephone wires stretching out to make numerous contacts with other neurons, and the tiny spines on these dendrites are responsible for receiving signals important for learning and memory. The study extends our prior findings that overexpression of PINK1 can increase branching of neurons and protect against the harmful dendritic shrinking effects of the parkinsonian toxins 6-OHDA and MPP+. By understanding better processes that trigger PINK1 degradation, we were able to develop a small molecule that elevates endogenous expression of PINK1 to achieve neuroprotection. These developments may lead to new therapies to help protect the ability of neurons to communicate through synaptic contacts. Aaron Gusdon, who just moved to Houston as an Assistant Professor of Neurosurgery at the UT Health Science Center, received notification that his K23 will be funded by NIH!
Aaron has a long term interest in mitochondrial pathobiology and will be studying metabolic effects of aneurysmal subarachnoid hemorrhages, while continuing to practice in Neuro-Critical Care. He joins an illustrious group of lab alumni who have achieved independent funding within two years of starting their first faculty position. Due to COVID-19, I had to deliver the Robbins Distinguished Educator Award Lecture via Zoom. |
Chu Lab
News and Press Releases Archives
September 2022
Categories
All
|







 RSS Feed
RSS Feed
